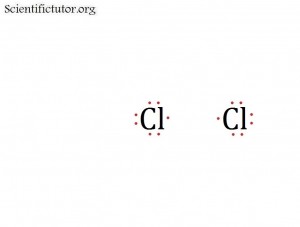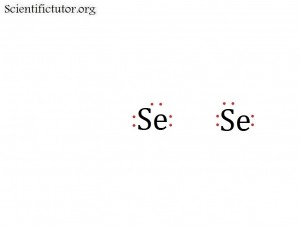Scientific Tutor
Covalent Bonds in Electron Dot Structures (Lewis Structures)
VIDEO explanation of the Covalent Bonds in an Electron Dot Structure (Lewis structure).
What is a covalent bond?
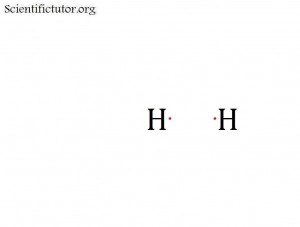
All following steps in creating and thinking about electron dot structures (Lewis structures) require that we first understand what a covalent bond is. When valence electrons from different atoms come near each other they can form covalent bonds by sharing their electrons in an orbital between them. That means the electrons travel back and fourth between the two atoms. The picture below represents two atoms with their valence electrons just before they are about to join in a covalent bond.
How do we represent covalent bonds in Electron Dot Structures (Lewis structures)?
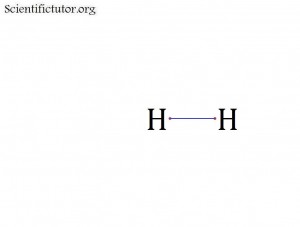
Each single covalent bond is made up of 2 electrons.
We can represent a covalent bond in an electron dot structure (Lewis structure) by a blue line that connects the 2 valence electron red dots in our pictures. An example is below.
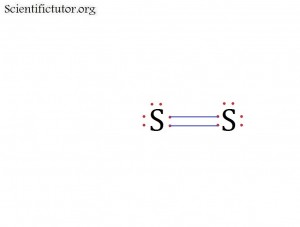
You can also create electron dot structures (Lewis structures) with more than one bond. This is known as creating double bonds or triple bonds. A double bond is just two bonds created between the same two atoms in an electron dot structure (Lewis structure). Example below.
A triple bond is just three bonds created between the same two atoms in an electron dot structure (Lewis structure). Example below.
PRACTICE PROBLEMS: Try drawing for yourself single, double, and triple bonds in structures. Start with the picture I have provided and then complete it.
Cl2 has a single bond between the two atoms.
Se2 has a double bond between the two atoms.
As2 has a triple bond between the two atoms.