Scientific Tutor
Chem – Electron Configuration Diagrams
What sections should I know before attempting to learn this section?
—> Complete Electron Configuration Part 3
—> Abbreviated Electron Configuration Part 4A
What are electron configuration diagrams?
VIDEO Explanation of electron configuration diagrams.
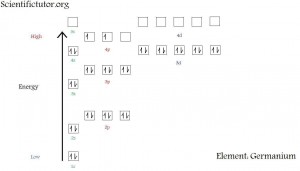
Electron configuration diagrams are how to represent the electron configurations in a picture form. Mainly what you need to know is how the picture below this paragraph is created. I will explain it in words as you are simultaneously looking at the picture below. You might also want to bring up the energy level periodic table. The electron configuration picture is displayed by putting the orbitals closest to the nucleus (of the lowest energy) further toward the bottom of the picture. As the orbitals get further from the nucleus (higher in energy) they are displayed higher and higher in the picture. For example, the 3p orbitals are higher on an electron configuration diagram then the 2s orbital. The orbital types (s, p, d, f…) are also organized in a specific way. All the s orbitals are lined up furthest toward the left side of the picture. The s orbitals are also stacked one on top of the next. The p orbitals are slightly to the right of the s orbitals and slightly above the s orbitals in the same energy levels. For example, the 2p orbital is to the right of and slightly higher than 2s orbitals. However, the p orbitals like the s orbitals, are stacked one on top of another.
What is each box representing in the picture above? Each box represents one orbital. Each orbital can only hold 2 electrons maximum. DO NOT CONFUSE THIS WITH THE ORBITAL TYPES. If you take the maximum number of electrons that an orbital type can hold and divide it by 2, you get the amount of orbitals in that orbital type. For example, the d orbitals can hold 10 electrons. So, 10 divided by 2 equals 5. That is why the picture above has 5 boxes representing the 3d orbitals. If we are only able to put 2 electrons in each of the d orbitals, then we can put a total of 10 electrons in all the 3d orbitals. In the electron configuration diagram above, the electrons are represented by the up and down arrows. It does not matter if it is an up or down arrow. Both of them represent an electron. The only difference between the up and down arrow is the direction the electrons are traveling. The up versus down arrows are saying that the electrons are traveling in opposite directions relative to one another. I describe this movement of electrons by comparing it to cars on a racetrack. If I were representing cars on a racetrack by arrows, I would make them go in all the same direction (all up), because all cars go in the same direction during the race. However, what if I took half the cars in my race and made them go in the other direction? Besides a lot of potential car accidents happening, you could then show the cars running the races as half of them being up arrows and half of them being down arrows. This is just like electrons which race toward each other within the same track (orbital). Although the electrons do not collide with each other because they are both negatively charged, the cars around a racetrack versus electrons in an orbital is a very close analogy to what is actually happening in an atom or orbital.
To determine which orbitals are closest to the nucleus (lowest in energy), you have to break down your determination into two categories. The first is what energy level is that orbital on. The second is what type of orbital is it. Orbitals of a lower energy level will be closer to the nucleus. For example, the 2p orbitals will be closer to the nucleus than the 4s orbitals because the 2p orbitals is on the second energy level and the 4s are on the fourth energy level. Also, there is an order of orbitals within the same energy level. The s orbitals will be closer to the nucleus than the p orbital within the same energy level. The p orbitals will be closer to the nucleus than the d orbital within the same energy level. The d orbitals will be closer to the nucleus than the f orbital within the same energy level. For example, the 3p orbitals are closer to the nucleus than the 3d orbitals.
Examples: Which orbitals are CLOSEST (HAVE THE LOWEST ENERGY) to the nucleus?
| 3s or 1s | 1s |
| 4f or 2p | 2p |
| 5d or 5f | 5d |
PRACTICE PROBLEMS: Which orbitals are CLOSEST (HAVE THE LOWEST ENERGY) to the nucleus?
| 5f or 4f | 4f |
| 7p or 6s | 6s |
| 2s or 2p | 2s |
| 4d or 4p | 4p |
| 3d or 4p | 3d |
| 6f or 6s | 6s |
Now that we have a general understanding of the number of orbitals, orbital types, and the relationship of orbitals to the nucleus we should be able to start constructing electron configuration diagrams. How do we do that? We should first open up a periodic table with the energy levels and orbital types clearly displayed. Here is the same periodic table viewed in a different way. Just like the electron configures before, we want to start any electron configuration by starting at the top of the periodic table in the 1s orbitals or the spot where hydrogen is. Since it is very hard to explain electron diagrams in text, there are video examples below.
VIDEO Example 1 Nitrogen: How to draw an electron diagram.
VIDEO Example 2 Sulfur: How to draw an electron diagram.
VIDEO Example 3 Iron: How to draw an electron diagram.
PRACTICE PROBLEMS: Draw the electron diagram of the following elements or charged atoms. Use this periodic table if possible. If that does not work for you try the orbital periodic table. It is best to draw them from memory but you can refer to a template electron diagram if you wish.
| B | Answer 1 link |
| F | Answer 2 link |
| Ca | Answer 3 link |
| Rh2+ | Answer 4 link |
| Se2- | Answer 5 link |
| Ru | Answer 6 link |