Scientific Tutor
Chem – Light and Electron Interactions
So what does the interaction of light and electrons look like?
Like light, electrons are also tiny particles. In addition, they move like light both very quickly and tend to follow wave patterns. What this means is that the particle of light, the photon, and the electron particle can interact. When these two particles (electrons and photons) interact, it turns out to be something like a pool (billiards) game. The photons can knock around the electrons and cause the electrons to move to different positions within an atom.
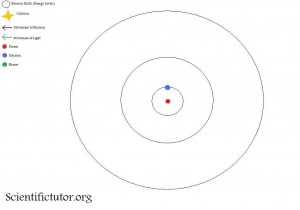
If we look at an atom as a Bohr model like the picture below, it allows us to gain a pretty easy understanding of the interactions between electrons and light. We will use the element hydrogen as our model atom. The first picture is what happens before the light particle (photon) hits the electrons. Notice how the electron starts on the closest electron shell (energy level) to the nucleus. The closest available shell (energy level) to the nucleus is where you will find the electron assuming no energy is put into it.
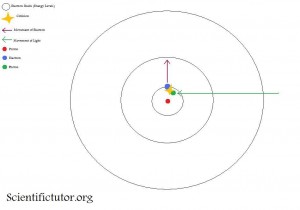
The second picture is what happens after the light particle (photon) hits the electron. See how the electron is moving from the first electron shell to the second electron shell. That was because the photon hit the electron and knocked or pushed it up one electron shell (energy level).
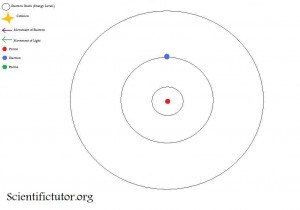
In the third picture, the electron has moved to the second shell.
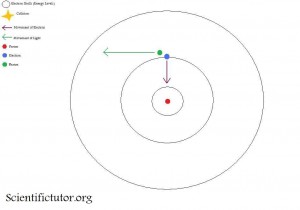
An instant (very very small) amount of time after the third, picture happens the fourth picture happens. The fourth picture is an electron falling back down to the closest shell to the nucleus while releasing a photon. It basically had this photon in storage and when it falls it releases it. Therefore, the light will travel away from the electron at this point.
In the fifth and last picture the electron in the hydrogen atom has returned to its original shell (energy level), closest to the nucleus.
This concept of electrons moving when they are struck by something else is actually the same basic concept that a soccer player uses to juggle a ball. Just like the soccer ball is being pulled to the Earth by gravity, the electron is being pull close to the nucleus because the electron (negatively charged) has an attraction to the protons (positively charged) in the nucleus. If you want to force the soccer ball away from the Earth, you have to knock (kick) your foot into the soccer ball. If you want to force the electron away from the nucleus, you have to knock the electron with a photon. When you knock the soccer ball, it flies away from the Earth and comes to some level for a split second and then falls back down. When you knock the electron, it flies away from the nucleus comes to a certain level for split second and then falls back down. You can even describe the energy that the electron stores as it rises and falls because light is a form of energy. The electron absorbs or releases that light energy as it changes position. Likewise, the soccer ball stores the energy in terms of sound. When you kick the soccer ball the collision of your foot and the ball make a sound. When the soccer ball falls back down it releases that sound once it hits something.
In fact, there is only one small difference between what happens with a soccer ball versus what happens with an electron. The electron follows a quantum (or is quantized) where as the soccer ball is not. Quantized means that the electron can only be at the first shell or the second shell or the third shell and so on. The electron cannot ever occupy the space in between the shells. So, if we run a video of the pictures in the example I gave you this is what it would look like. You see the electron at the first shell. You see the electron disappear. You see the electron reappear at the second shell. You see the electron disappear. You see the electron reappear at the first shell. It is kind of strange to think about but that is how it works. On the other hand, the soccer ball can move and occupy any space in between its starting point and its’ highest point after it is kicked by a person.
VIDEO Here is a video demonstration of the electron versus the football (soccer ball).
During the electron’s travel between different energy levels (shells), you can see different wavelengths of light coming off of the atom. The WAVELENGTH of light that the electron gives off when it falls back from a higher energy level to a lower energy level depends on the DISTANCE traveled by that electron. The GREATER the distance the HIGHER the energy, the HIGHER the frequency, and therefore the SHORTER the wavelength. For example, in most atoms, if an electron jumps only one energy level (shell) you will see a long wavelength (red side of the spectrum). However, if an electron jumps 4 or more energy levels then you will see a very short wavelength (purple side of the spectrum).