Scientific Tutor
Chem – Bond Angle
What is the bond angle?
Bond angle is another way to look at the molecular shapes or 3-D structure of a flat 2-D drawn electron dot structure (Lewis structure). It is the angle between any two covalent bonds in an electron dot structure.
How do electron dot structures (Lewis structures) relate to the bond angle?
Bond angle and molecular shape are essentially the same thing, but we are only talking about it from a different perspective. The bond angle is the shortest angle between one bond and another bond. This again is driven by the electrons in the bonds and lone pairs of the central atom repelling each other and trying to maintain the furthest distance apart as possible. The theory of electrons repelling each other to create the shape of the molecule is called the VSEPR (Valence Shell Electron Pair Repulsion) model.
How do you determine the bond angle from the electron dot structure (Lewis structure)?
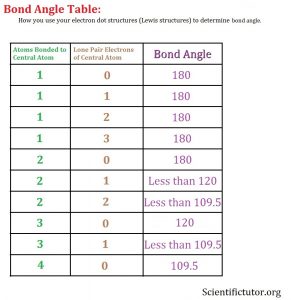
What you are going to focus on to determine the bond angle of a molecule is the central atom from your electron dot structure. You are going to ask the question, HOW MANY ATOMS ARE ATTACHED TO THE CENTRAL ATOM through bonds AND HOW MANY LONE PAIRS EXIST AROUND ONLY THE CENTRAL ATOM? After that you can refer to the bond angle table below:
Once you add up the number of atoms bonded to the central atom and the number of lone pair electrons around the central atom as two distinct and separate numbers, then you will get your look over at the bond angle column and get your answer.
Examples: Use the electron dot structure (Lewis structure) and the bond angle table to determine the bond angle. VIDEO Bond Angle from Lewis Structure Examples 1.
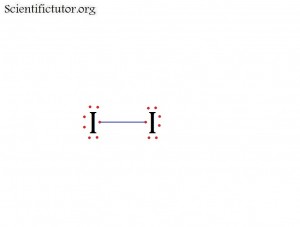
Atoms bonded to central atom: 1
Lone pair electrons of central atom: 3 pairs (6 electrons total)
Bond Angle of I2 (Answer): 180 degrees
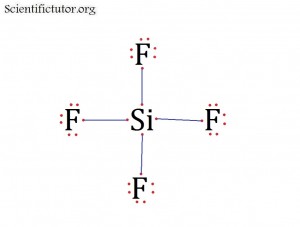
Atoms bonded to central atom: 4
Lone pair electrons of central atom: 0 pairs
Bond Angle of SiF4 (Answer): 109.5 degrees
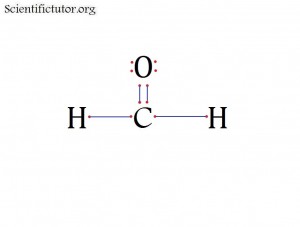
Atoms bonded to central atom: 3
Lone pair electrons of central atom: 0 pairs
Bond Angle of H2CO (Answer): 120 degrees
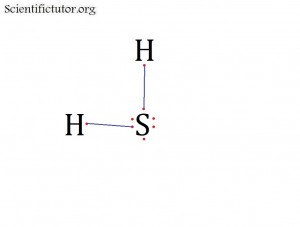
Atoms bonded to central atom: 2
Lone pair electrons of central atom: 2 pairs (4 electrons total)
Bond Angle of SH2 (Answer): Less than 109.5 degrees
PRACTICE PROBLEMS: Use the electron dot structure (Lewis structure) and the bond angle table to determine the bond angle.
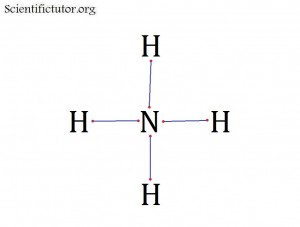
Atoms bonded to central atom: 4
Lone pair electrons of central atom: 0 pairs
Bond Angle of NH4+ (Answer): 109.5 degrees
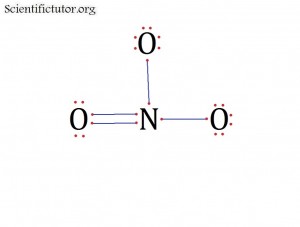
Atoms bonded to central atom: 3
Lone pair electrons of central atom: 0 pairs
Bond Angle of NO31- (Answer): 120 degrees
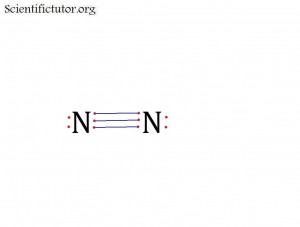
Atoms bonded to central atom: 1
Lone pair electrons of central atom: 1 pair
Bond Angle of N2 (Answer): 180 degrees
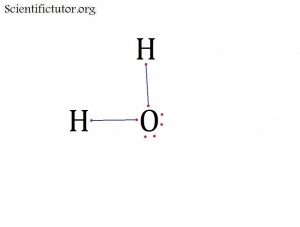
Atoms bonded to central atom: 2
Lone pair electrons of central atom: 2 pairs
Bond Angle of H2O (Answer): Less than 109.5 degrees
Atoms bonded to central atom: 3
Lone pair electrons of central atom: 1 pair
Bond Angle of PCl3 (Answer): Less than 109.5 degrees
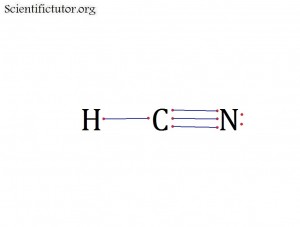
Atoms bonded to central atom: 2
Lone pair electrons of central atom: 0 pairs
Bond Angle of HCN (Answer): 180 degrees